Since the principal instance of COVID-19 in the UAE, in late January, the nation has driven a quick reaction to the novel coronavirus pandemic.
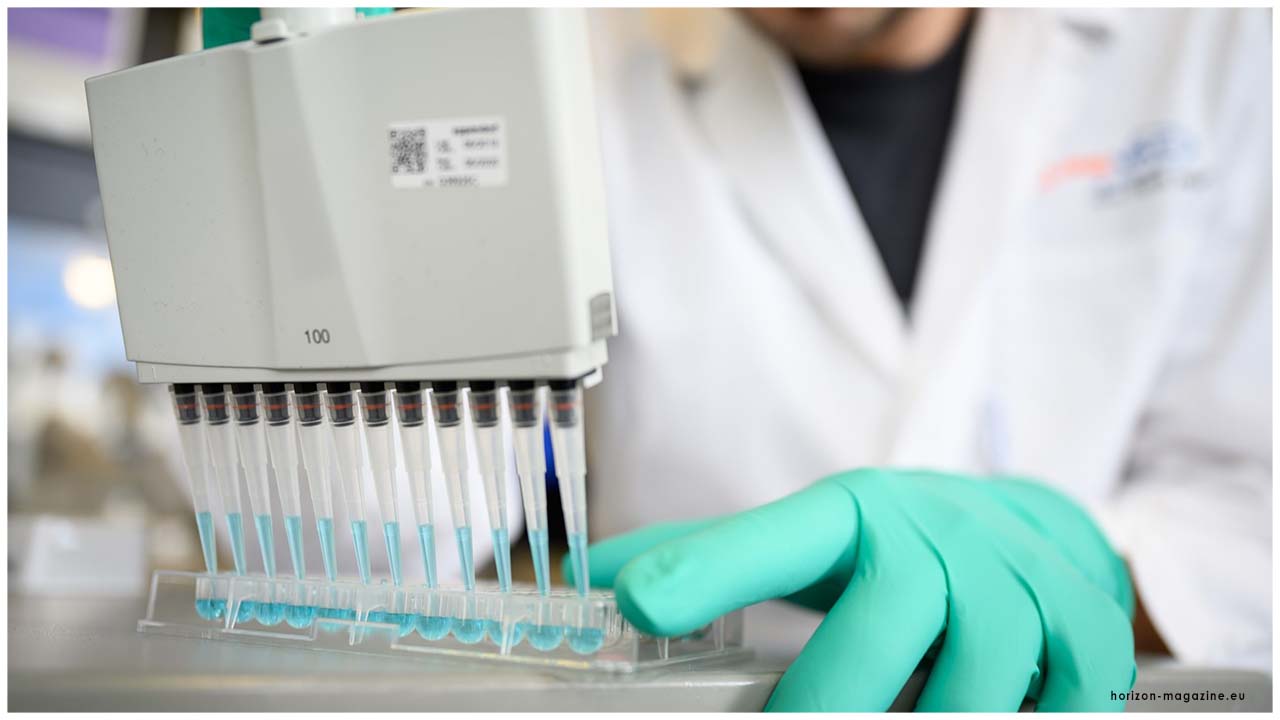
Masdar City in Abu Dhabi ventured up to turn into an inside for high-volume testing, the first outside of China, with an ability to test a huge number of individuals every day.
Until this point in time, the UAE has one of the most noteworthy testing rates for COVID-19 on the planet with in excess of 4,000,000 tests led by the UAE Ministry of Health and Prevention.
Solidarity and improvement
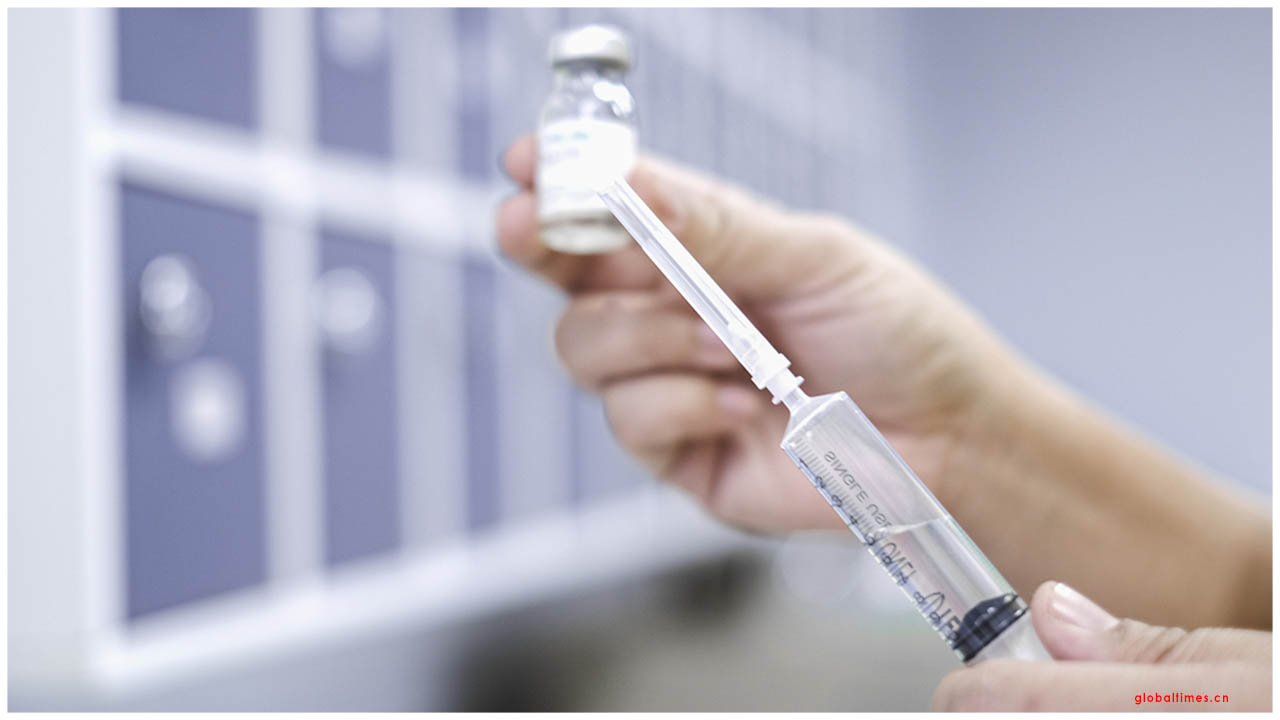
Wellbeing experts in Abu Dhabi and Beijing as of late reported an organization between the Chinese pharmaceutical organization Sinopharm, and Group 42, the Abu Dhabi-based man-made consciousness and distributed computing organization.
The two accomplices state this will commence the start of stage III clinical preliminaries for a COVID-19 immunization, to test whether it is successful and safe to utilize.
These clinical preliminaries were affirmed by Dr. Abdul Rahman canister Mohammed receptacle Nasser Al Owais, the UAE Minister of Health and Prevention.
Numerous American and European organizations are additionally creating possible antibodies, Euronews asked Ashish Koshy, the CEO of Group 42 Healthcare, why an association with China is the best fit for the UAE.
"Sinopharm has two items, two inactivated immunizations in the best ten race," says the CEO. "What's more, that is the fundamental cooperative energy we saw that felt like a very solid accomplice that we could bring into UAE, acquire the ability to the UAE, and give early access to the UAE occupants."
Worldwide race
Different governments and associations around the globe are putting assets in the disclosure of a potential antibody for COVID-19, in any case, Koshy accepts that the quest for a fix isn't a race.
"It's a worldwide exertion together against this worldwide pandemic. Each nation needs to have its impact in taking an interest," he discloses to Inspire Middle East's Rebecca McLaughlin-Eastham.
Gathering 42 is as of now sequencing COVID-19 in their genomics research center with the objective of understanding the immunization's effect on people and the infection the same
The organization has likewise built up an AI stage to recognize designs in the infection's change during stage III preliminaries and decide its viability.
"We have plenty of information where individuals can connect and think of amazing bits of knowledge that could make sense of how the infection transforms and in this way, conceivably, the antibody with a response to fix that COVID-19," clarifies Koshy.
Future possibilities
The cooperative immunization has just passed stages I and II. Stage II preliminaries finished up with 100 percent of volunteers creating antibodies following two dosages in 28 days, as indicated by Koshy.
Koshy affirms that now in stage III, the organization is focusing on a gathering of somewhere in the range of 5,000 volunteers to test the antibody in the UAE.
Whenever affirmed protected and viable, the medication will go for the Food and Drug Administration (FDA) endorsement, and if positive, the organization will enter the last enormous scope fabricating stage.
The organizations included are confident that immunization may enter the market before the finish of 2020 or mid-2021.
At present, the UAE has enlisted around 55,000 instances of COVID-19 as it proceeds with its endeavors to battle the pandemic.

 Masdar City in Abu Dhabi stepped up to become a centre for high-volume testing, a first outside of China.
Masdar City in Abu Dhabi stepped up to become a centre for high-volume testing, a first outside of China.