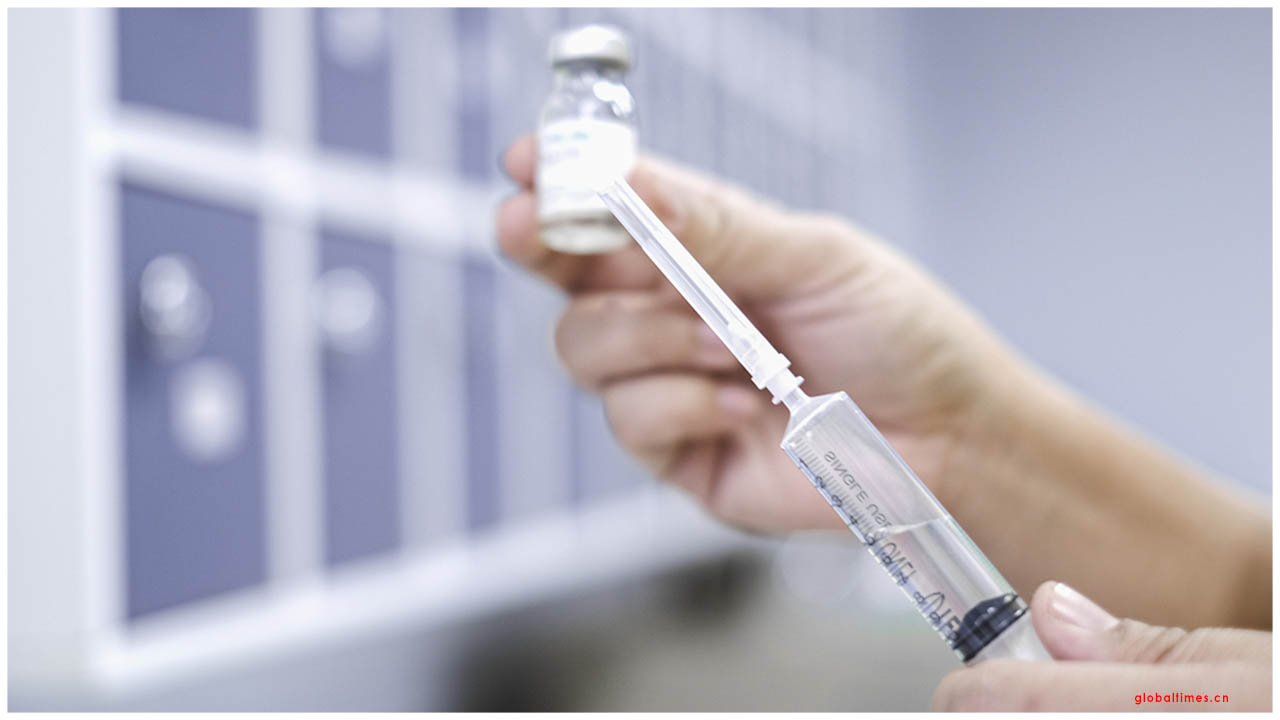
The crisis authorization of COVID-19 vaccination requires a "lot of earnestness and reflection", the World Health Organization said on Monday after the United States declared it was thinking about optimizing up-and-comer drugs.
Albeit each nation reserved the privilege to affirm drugs without finishing full preliminaries, "it isn't something that you do daintily", WHO boss researcher Soumya Swaminathan told newsgathering.
The top of the U.S. Food and Drug Administration said he would sidestep the typical endorsement cycle to approve a COVID-19 immunization as long as authorities were persuaded the advantages exceed the dangers.
Russia has just allowed administrative endorsement to a COVID-19 immunization this month after under two months of human testing, provoking some Western specialists to scrutinize its security and viability.
The WHO's favored methodology is to have a full arrangement of information that could be utilized for the pre-capability of immunizations, Swaminathan said. The WHO might then think about the adequacy and wellbeing of each medication depending upon the situation, she included.
The WHO has utilized exploratory medications to battle Ebola in Africa, a measure which demonstrated fruitfully, Mike Ryan, the top of the association's crises program, said.
However, he focused on that a most optimized plan of attack approach without full preliminaries required serious checking and security follow-up work, and ought to be ended promptly if issues happen.
'If you move too quickly to vaccinate millions of people, you may miss certain adverse effects," Ryan said

 The head of the U.S. Food and Drug Administration said he would be willing to bypass the normal approval process to authorise a COVID-19 vaccine
The head of the U.S. Food and Drug Administration said he would be willing to bypass the normal approval process to authorise a COVID-19 vaccine