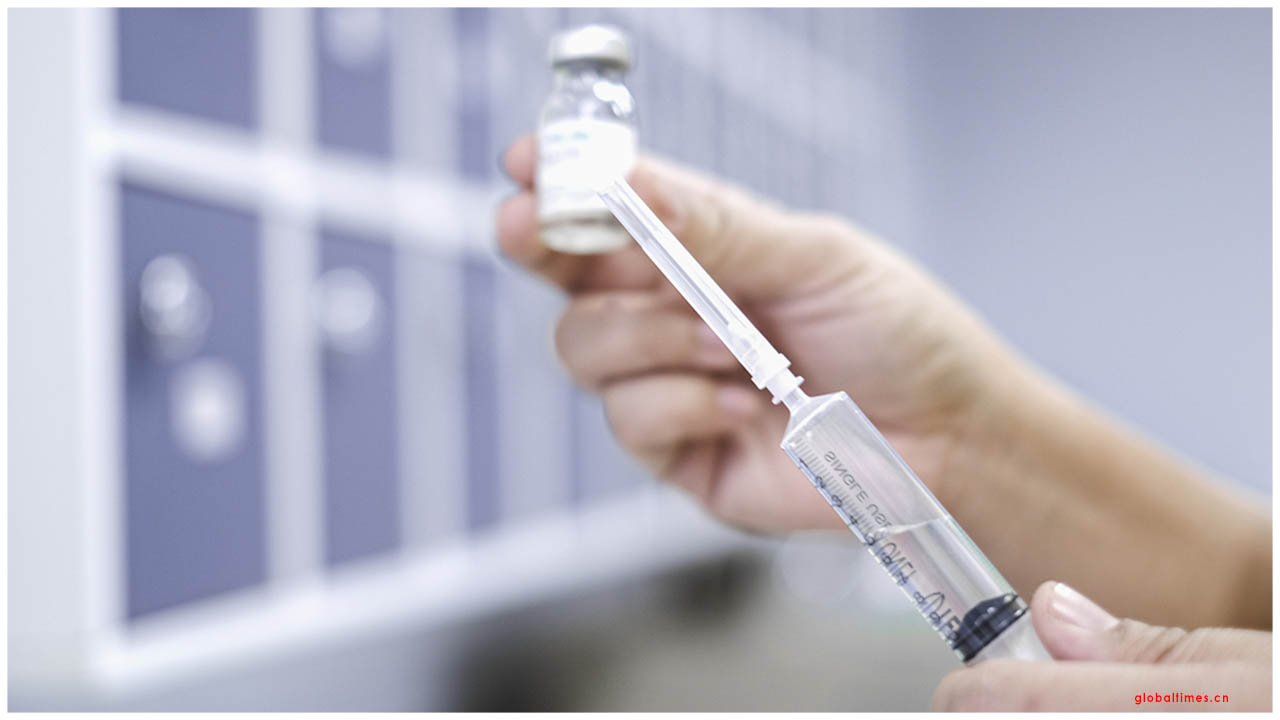
Gilead Sciences Inc on Monday evaluated its COVID-19 antiviral remdesivir at $2,340 per quiet for wealthier countries and consented to send almost the entirety of its flexibly of the medication to the United States throughout the following three months.
The sticker price is marginally beneath the scope of $2,520 to $2,800 recommended a week ago by the U.S. medicate evaluating research bunch the Institute for Clinical and Economic Review (ICER) after British specialists said they found that the modest, generally accessible steroid dexamethasone fundamentally decreased mortality among seriously sick COVID-19 patients.
Remdesivir is relied upon to be sought after as one of the main medicines so far appeared to change the course of COVID-19. After the intravenously managed medication abbreviated emergency clinic recuperation times in a clinical preliminary, it won crisis use approval in the United States and full endorsement in Japan.
The medication is accepted to be best in rewarding patients before throughout infection than dexamethasone, which diminished passings in patients requiring steady oxygen and those on a ventilator. All things considered, remdesivir in its at present detailing, is just being utilized on patients sufficiently debilitated to require hospitalization as a five-day treatment course.
The organization is building up a breathed in a form that could be utilized outside a clinic setting.
For U.S. patients with business protection, Gilead said it will charge $3,120 per course or $520 per vial. That is a 33% expansion over the $390 per vial Gilead said it will charge legislatures of created nations and U.S. patients in government social insurance programs.
'Preposterous Price For A Very Modest Drug'
In an open letter, Gilead Chief Executive Daniel O'Day said the cost is well underneath the worth it gives given that early medical clinic releases could spare around $12,000 per quiet in the United States.
Quiet backers have contended that the expense ought to be lower since remdesivir was created with budgetary help from the U.S. government.
U.S. Agent Lloyd Doggett, a Democrat from Texas, said it was "a crazy cost for a humble medication, which citizen subsidizing spared from a garbage dump of disappointments."
Remdesivir had recently fizzled as an Ebola treatment and has not demonstrated that it can lessen COVID-19 passings.
Gilead likewise said it consented to keep on sending the greater part of its flexibly of remdesivir to the U.S. Branch of Health and Human Services (HHS), with the organization and states set to oversee assignment to U.S. medical clinics until the finish of September.
There are at present more instances of COVID-19 in the United States than in Europe, with a few U.S. states hitting new records for quantities of cases.
HHS has been conveying the medication since May and was because of running out after this week. A senior HHS official said the organization expects the medication will before long be a scant asset, thus it needed to stay associated with assigning it.
The organization said it made sure about more than 500,000 remdesivir courses for U.S. emergency clinics through September. That speaks to the entirety of Gilead's anticipated creation for July and 90% of its creation in August and September, notwithstanding a distribution for clinical preliminaries, HHS said.
When supplies are less compelled, HHS will quit dealing with the distribution, Gilead said. The organization didn't examine its graceful technique for created countries outside the United States.
Remdesivir's cost has been a subject of extraordinary discussion. Specialists have said Gilead would need to abstain from seeming to exploit a wellbeing emergency for benefits.
Gilead shares were about level on Monday.
Examiners at Royal Bank of Canada conjecture the medication could create $2.3 billion in income 2020, helping balance more than $1 billion being developed and circulation costs. They said extra benefits could be constrained because antibodies and better medicines are not too far off.
The European Union's social insurance controller a week ago suggested contingent endorsement of the medication when utilized in the fundamentally sick.
Gilead has connected up with nonexclusive drugmakers situated in India and Pakistan, including Cipla Ltd and Hetero Labs Ltd, to make and gracefully remdesivir in 127 creating nations.
Cipla's adaptation is valued at under 5,000 Indian rupees ($66.24), while Hetero Lab's form is evaluated at 5,400 rupees ($71.54).

 Remdesivir is expected to be in high demand as one of the only treatments so far shown to alter the course of COVID-19.
Remdesivir is expected to be in high demand as one of the only treatments so far shown to alter the course of COVID-19.